 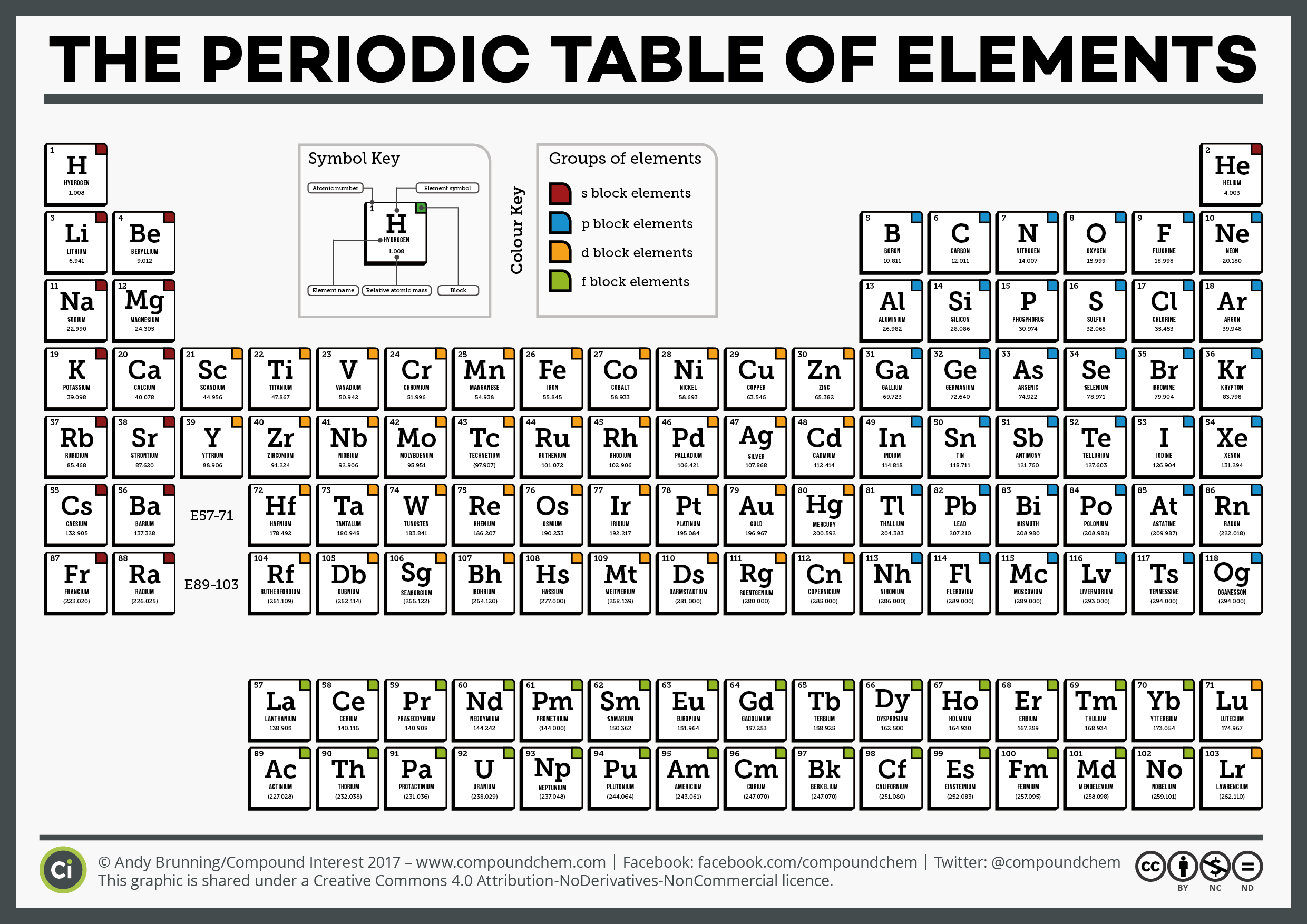
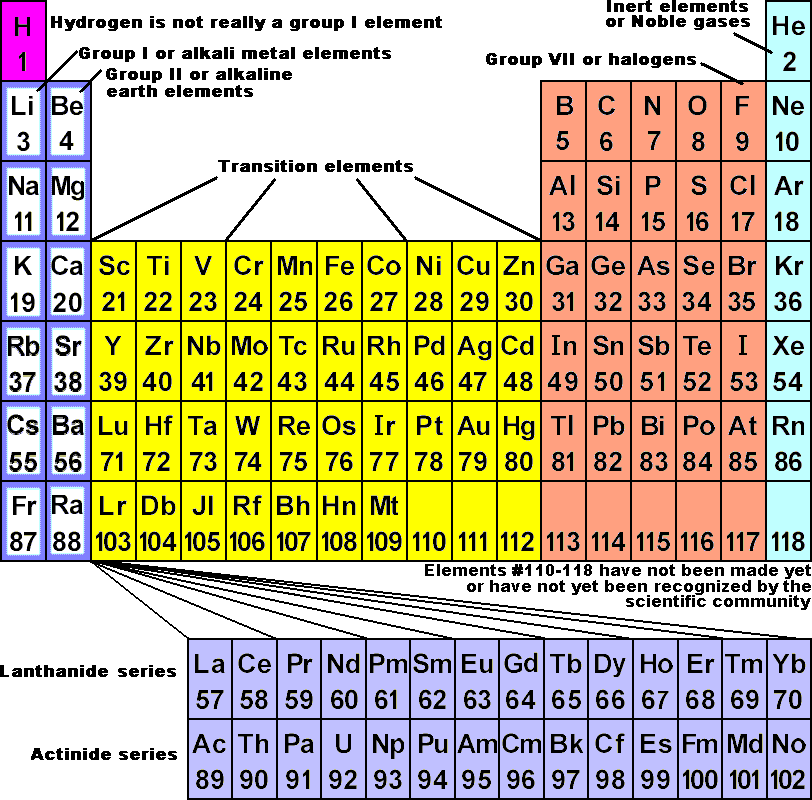
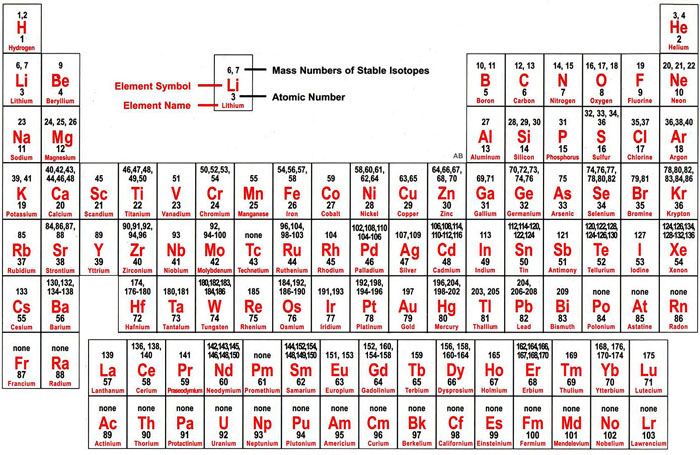
In other words, even though an element group might break a trend, the elements within the group display periodic properties. However, the behavior of the noble gases is periodic. The noble gases are an exception to the trend since these elements have filled electron valence shells and electron affinity values approaching zero. Nonmetals usually have higher electron affinities than metals. Electron affinity increases moving across a period and decreases moving down a group.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
